BERLIN (Reuters) -German pharma company Bayer reported positive study results for its anticoagulant asundexian on Sunday, two years after a research setback for the promising blood thinner candidate.
In a Phase III study, a daily dose of 50 milligrams significantly reduced the risk of ischemic stroke compared with a placebo, Bayer said.
Detailed results from the OCEANIC-STROKE study will be presented at an upcoming scientific congress, said Bayer.
Bayer added that it plans to speak with health authorities worldwide in preparation for the submission of marketing authorisation applications.
Bayer had originally predicted that asundexian would have peak sales potential of more than 5 billion euros ($5.76 billion) - more than any of its other drugs.
At the end of 2023, the company had a major setback with the drug after it failed in a pivotal clinical trial involving patients with atrial fibrillation and a risk of stroke.
($1 = 0.8687 euros)
(Reporting by Joern Poltz. Writing by Miranda Murray. Editing by Jane Merriman)
latest_posts
- 1
 Amy Poehler's podcast is a hit. It's also a Trojan horse for talking about women and aging.
Amy Poehler's podcast is a hit. It's also a Trojan horse for talking about women and aging. - 2
 January full moon wows skywatchers with a striking 'Wolf Supermoon' (photos)
January full moon wows skywatchers with a striking 'Wolf Supermoon' (photos) - 3
 Watch China's Shenzhou 22 rescue ship arrive at Tiangong space station (video)
Watch China's Shenzhou 22 rescue ship arrive at Tiangong space station (video) - 4
 Figure out How to Streamline Eco-friendliness in Your Volvo XC40
Figure out How to Streamline Eco-friendliness in Your Volvo XC40 - 5
 Aspect Biosystems receives funding for cellular medicine project
Aspect Biosystems receives funding for cellular medicine project
 More parents refusing this shot that prevents serious bleeding at birth
More parents refusing this shot that prevents serious bleeding at birth Holyvolt And Wildcat Could Help The West Reclaim Battery Leadership
Holyvolt And Wildcat Could Help The West Reclaim Battery Leadership This Week In Space podcast: Episode 192 — Space, 2026!
This Week In Space podcast: Episode 192 — Space, 2026! How federal officials talk about health is shifting in troubling ways – and that change makes me worried for my autistic child
How federal officials talk about health is shifting in troubling ways – and that change makes me worried for my autistic child A Couple of Modest Guitars for 2024
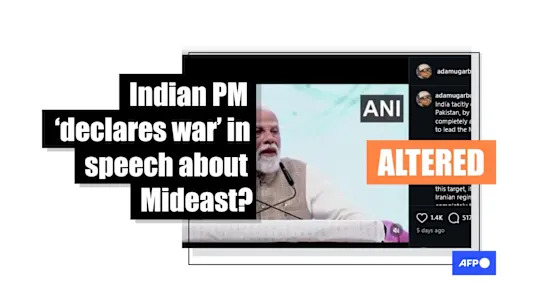
A Couple of Modest Guitars for 2024 Doctored NXT Summit footage falsely portrays Modi as declaring war on Iran and Pakistan
Doctored NXT Summit footage falsely portrays Modi as declaring war on Iran and Pakistan More charges filed against ex-left-wing RAF member Daniela Klette
More charges filed against ex-left-wing RAF member Daniela Klette If evolution is real, then why isn’t it happening now? An anthropologist explains that humans actually are still evolving
If evolution is real, then why isn’t it happening now? An anthropologist explains that humans actually are still evolving Discovering a sense of harmony: Individual Accounts of Reflection and Care
Discovering a sense of harmony: Individual Accounts of Reflection and Care